 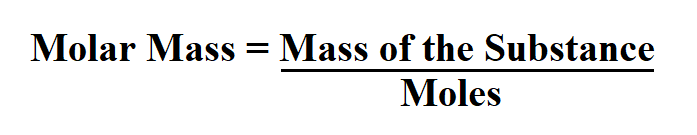
Using our Molecular Weight Calculator we can easily get 30.026 g/mol. To this end, we first find the molecular weight of the empirical formula, that is, the weight in grams of one mole of a substance having the chemical formula CH 2O. Now, if we know that the molar mass of the compound under study is 180.16 g/mol, we can find the molecular formula of this compound. If we now divide the found numbers of moles by the minimum value of 0.1665 and round to whole values, then we get the following indices for the empirical formula: 1, 2, 1. The molecular formula of a substance is always the empirical formula of that substance whose indices are all multiplied by the same integer n, which can be symbolically expressed as the following formula:Ġ.336 g H / 1.008 (g/mol) H = 0.3333 mol H Then we consider the number of moles of each element relative to each other, converting these numbers into a whole-number ratio that can be used to derive the empirical formula of the substance. The easiest way to express the number of atoms is in moles.įor this, we have to use molar masses to convert the mass of each element to a number of moles. So, any experimental data involving masses must be used to derive the corresponding numbers of atoms in the compound. The most common approach to determining a compound’s chemical composition is to measure first the masses of its constituent elements. To find the empirical formula of a substance you can use our Empirical Formula Calculator. The empirical formula of a chemical compound is the simplest whole number ratio of atoms present in the compound. The molecular formula should be distinguished from the empirical formula.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
